![[RFE/RL]Appeal Grows For Russian, Chinese Serums As Western Vaccine Efforts Get Bogged Down[Pakistan News Updates] [RFE/RL]Appeal Grows For Russian, Chinese Serums As Western Vaccine Efforts Get Bogged Down[Pakistan News Updates]](https://gdb.rferl.org/5430afba-073a-43f1-8dd5-4eb44ef6fa63_cx0_cy9_cw100_w250_r1_s.jpg)
A new study has helped Russia’s leading coronavirus vaccine bounce back from criticism of a premature rollout just as many countries are confronting a growing array of logistical and political challenges to delivering vaccines.
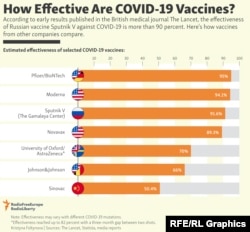
The interim analysis of the Sputnik V’s Phase 3 trial results, which were published on February 2 in the international medical journal The Lancet, found that Sputnik V is 91.6 percent effective against symptomatic COVID-19 and effective against severe and moderate disease.
That is roughly comparable to the protection conferred by the leading Western-made vaccines so far.
While the research published is unclear over whether the Russian injection can stop transmission of the virus, the new study is already tempering some concerns over a lack of transparency and a hasty initial rollout before clinical trials were completed.
“There are issues about the speed and how it was pushed out that caused some concern, but this new dataset from The Lancet should neutralize those concerns,” Julian Tang, a clinical virologist at the University of Leicester in the United Kingdom, told RFE/RL. “Russia bet on its vaccine and that bet seems to have paid off.”
The new study comes as the race to produce effective and easy-to-distribute vaccines against COVID-19 enters a new stage.
Western vaccines from AstraZeneca, Moderna, and Pfizer/BioNTech, along with China’s Sinovac and Sinopharm and Russia’s Sputnik V, have all emerged as early winners.
But national governments’ fresh jockeying for limited supplies has brought on a host of mostly foreseeable problems — from production and delivery delays to distribution challenges — that appear to be encouraging a higher tolerance for opacity and risk.
This has carved out a greater global foothold for the Russian and Chinese vaccines despite some remaining questions over the injections.
“China and Russia put their feet on the gas with [domestic] approval of their vaccines prematurely,” Judy Twigg, a global-public-health expert at Virginia Commonwealth University, told RFE/RL. “One of the things they’re hoping to do is take advantage of the void that has been created by the vaccine nationalism that we’ve seen in the West.”
Bureaucratic infighting appears to have slowed the European Union’s rollout, and Brussels and AstraZeneca are locked in a dispute after the pharmaceutical giant said it would have to scale back deliveries because of a manufacturing issue. The European Union has insisted that the drugmaker honor its contract and has since enacted export controls on vaccines produced within the bloc.
Meanwhile, there is little sign that developing countries will get comparable access anytime soon. While countries in parts of Eastern Europe, Central Asia, the Middle East, and South Asia prepare their vaccination programs, rollouts are expected to be slow, with a lack of access to injections a major issue.
Many of those nations are participating in a World Health Organization (WHO)-led program aimed at equitable access, called COVAX, that has raised $2 billion to buy vaccines for poorer countries.
But getting access to enough doses for their populations could take years. A paper published in late January by the Economist Intelligence Unit said those regions were unlikely to see widespread vaccine coverage until 2023.
“There’s a vaccine vacuum, both in terms of public health, but also politically, and it’s one that Russia and China are more than happy to try and fill,” said Twigg.
Filling The Vacuum
Despite overriding questions early on, demand for Sputnik V is increasing.
The Russian vaccine has so far been approved by dozens of countries around the world for direct purchase or through licensing deals to their production.
“Russia certainly didn’t do itself any favors in the credibility department by having approved the vaccine before they had even launched Phase-3 trials. It’s their own action that has created the skepticism around the vaccine that we’ve seen for months,” said Twigg. “But it’s hard to imagine that they’ve just blatantly falsified the data that they sent to The Lancet. Results don’t make it into The Lancet without careful peer review by international teams of specialists.”
Countries like Belarus, Kazakhstan, and Turkey have already signed up to manufacture and develop Sputnik V, with Iran, which has banned Western vaccines, also agreeing to join. India, South Korea, Brazil, and China will also produce it, according to the Russian Direct Investment Fund, which funded the vaccine and is responsible for selling it globally.
WATCH: More Countries Look To Produce Russia’s Sputnik Vaccine As U.K. Journal Deems It Safe, Effective
When Russia granted the vaccine the regulatory go-ahead six months ago and launched mass vaccinations before data from Phase 3 trials was unveiled, scientists expressed concerns and lamented the lack of transparency. But the latest Phase 3 dataset for Sputnik V, along with the vaccine’s relatively low cost and simpler storage requirements is sure to boost its appeal across Eurasia.
Unlike the Pfizer and Moderna vaccines, Sputnik V and the two leading Chinese vaccines do not need to be stored and transported in extreme cold, giving them a logistical advantage for poorer countries that often lack facilities.
The Russian and Chinese injections may be the earliest options for nations at the back of the line for other vaccines.
Some Central Asian governments are counting on the COVAX program to inoculate a critical mass of their citizens, but the program limits itself to vaccinating 20 percent of any participating country’s population.
In order to bridge the gap, both Kyrgyzstan and Tajikistan have asked Russia for assistance in procuring Sputnik V doses; Turkmenistan, which claims to be coronavirus-free, has already begun to purchase Russian vaccines, according to RFE/RL’s Turkmen Service.
In addition to relying on COVAX, Uzbekistan began Phase 3 clinical trials in December 2020 for another Chinese vaccine developed by the company Anhui Zhifei Longcom Biopharmaceutical.
Pakistan, meanwhile, has received a donation of 500,000 Sinopharm vaccines from Beijing for a country of around 220 million. In addition to Sinopharm, Islamabad has already granted emergency-use authorization to the AstraZeneca and Sputnik V vaccines.
The Russian and Chinese vaccines are also gaining traction in Europe.
German Chancellor Angela Merkel said that Sputnik V could be used in the European Union if it got regulatory approval. German Health Minister Jens Spahn has hinted that coronavirus vaccines from China and Russia could be used to overcome Europe’s current deficit of doses.
EU member Hungary has already approved the Sinopharm vaccine and received its first shipment of Sputnik V — 40,000 doses — on February 2. On the EU’s periphery, Serbia has managed to vaccinate the first of its 7 million residents at Europe’s second-fastest rate, trailing the United Kingdom, by relying heavily on Russian and Chinese vaccines in addition to some Western doses.
Final Concerns In Vaccine Push
While The Lancet’s endorsement has calmed some concerns about Sputnik V’s quick approval process, an absence of similar studies of Chinese-made vaccines invites questions about such inoculations’ value. A study based on Sinovac data was published in The Lancet, but it only included information for Phase 1 and 2 trials.
Neither Sinovac nor Sinopharm has made its Phase 3 data public or published results, although both have submitted data to the WHO for review and received regulatory approval in other countries, as well as within China, based on their trials.
So far, Sinovac seems to be on track, although it has yielded varying levels of effectiveness in late-stage trials. Interim data from Turkey and Indonesia showed that the vaccine was 91.25 percent and 65.3 percent effective, respectively. Researchers in Brazil initially said it was 78 percent effective in their clinical trials, but revised that figure to 50.4 percent in January after including more data.
“There is still a lack of data for the Chinese vaccines,” said Twigg. “As much as there has been a lack of transparency from Russia with its vaccine initially, there has been even less transparency from China.”
In late December 2020, Sinopharm said that interim Phase 3 trials of the vaccine showed that it was 79 percent effective — lower than the Pfizer and Moderna shots but still considered above a strong threshold. But no complete dataset similar to that cited by The Lancet for Sputnik V has emerged for either of the Chinese vaccines.
While many experts note that the speed and lack of transparency around the test data of the Chinese vaccines have fueled skepticism over its effectiveness, preliminary trial results have so far given no cause for alarm.
“It’s difficult to separate the vaccine from the country it came from,” said the University of Leicester’s Tang. “But take a step back and look purely at the science and there are no significant adverse effects that I can see.”
[#Pakistan News Update]
Source link
Copyright (c) 2019. RFE/RL, Inc. Reprinted with the permission of Radio Free Europe/Radio Liberty, 1201 Connecticut Ave NW, Ste 400, Washington DC 20036.
![[Daily Excelsior]India, US regularly consult at highest levels on democracy, human rights issues: Official [Daily Excelsior]India, US regularly consult at highest levels on democracy, human rights issues: Official](https://cdndailyexcelsior.b-cdn.net/wp-content/uploads/2023/12/human-rights-696x392.jpg)
![[Times of India] PM Modi's salvos likely part of bigger attack on Congress manifesto's 'equity' section | India News – Times of India [Times of India] PM Modi's salvos likely part of bigger attack on Congress manifesto's 'equity' section | India News – Times of India](https://static.toiimg.com/photo/msid-109542445,imgsize-828744.cms)

![[The Hindu]J&K police announce ₹10 lakh on terrorist behind Rajouri killing [The Hindu]J&K police announce ₹10 lakh on terrorist behind Rajouri killing](https://www.thehindu.com/theme/images/og-image.png)
![[Indian Express]India 4th largest military spender in 2023: SIPRI [Indian Express]India 4th largest military spender in 2023: SIPRI](https://data.indianexpress.com/election2019/track_1x1.jpg)
![[Saudigazette]Modi: India’s prime minister eyeing a historic third term [Saudigazette]Modi: India’s prime minister eyeing a historic third term](https://saudigazette.com.sa/uploads/images/2024/04/23/2379093.jpg)
![[Rising Kashmir]“Cradled Art 370 like a baby for 70 years”: Amit Shah slams Cong [Rising Kashmir]“Cradled Art 370 like a baby for 70 years”: Amit Shah slams Cong](https://img.risingkashmir.com/wp-content/uploads/2024/04/20113232/20240416105L.jpg)
![[Deccan Chronicle]India’s Problems Began in Nehru Era, Says Jaishankar [Deccan Chronicle]India’s Problems Began in Nehru Era, Says Jaishankar](https://www.deccanchronicle.com/h-upload/2024/04/23/1085699-pav1854.webp)
![[The Northlines]PDP Has Become BJP’s C-Team, Says Omar Abdullah [The Northlines]PDP Has Become BJP’s C-Team, Says Omar Abdullah](https://thenorthlines.com/wp-content/uploads/2024/04/OMAR-5-1024x563-1.jpg)
![[The Northlines]Terror module member handled by Pak-based operatives held in Punjab [The Northlines]Terror module member handled by Pak-based operatives held in Punjab](https://thenorthlines.com/wp-content/uploads/2023/08/Northlines-01-300x300-1-1-1.png)
![[Rising Kashmir]PDP has emerged as ‘C Team’ of BJP: Omar Abdullah [Rising Kashmir]PDP has emerged as ‘C Team’ of BJP: Omar Abdullah](https://img.risingkashmir.com/wp-content/uploads/2024/04/23213654/images-14-2.jpeg)
![[Daily Excelsior]J&K Police Announces Rs 10 Lakh Reward For Terrorist Involved In Rajouri Killing [Daily Excelsior]J&K Police Announces Rs 10 Lakh Reward For Terrorist Involved In Rajouri Killing](https://cdndailyexcelsior.b-cdn.net/wp-content/uploads/2024/04/JK-POLICE-2-3-1-1-696x398.png)

![[RFE/RL]Iranian President Holds Talks With Pakistani PM In First Meeting Since Cross-Border Strikes[Pakistan News Updates] [RFE/RL]Iranian President Holds Talks With Pakistani PM In First Meeting Since Cross-Border Strikes[Pakistan News Updates]](https://gdb.rferl.org/01000000-0aff-0242-a566-08dc58668c66_w250_r0_s.jpg)
![[RFE/RL]Iran's President Raisi To Make Official Visit To Pakistan This Week[Pakistan News Updates] [RFE/RL]Iran's President Raisi To Make Official Visit To Pakistan This Week[Pakistan News Updates]](https://gdb.rferl.org/d3f182c9-154e-421a-9605-831ceb36f722_w250_r1_s.jpg)
![[RFE/RL]Latest Attack On Pakistani Customs Officials Kills 3[Pakistan News Updates] [RFE/RL]Latest Attack On Pakistani Customs Officials Kills 3[Pakistan News Updates]](https://gdb.rferl.org/01000000-0aff-0242-1cfa-08dc297e7d3d_w250_r0_s.jpg)
![[RFE/RL]Japanese Nationals Escape Suicide Attack In Pakistan[Pakistan News Updates] [RFE/RL]Japanese Nationals Escape Suicide Attack In Pakistan[Pakistan News Updates]](https://gdb.rferl.org/01000000-0aff-0242-86fb-08dc60708466_w800_h450.jpg)
![[RFE/RL]The Azadi Briefing: Taliban Deals Another Blow To Afghan Media[Pakistan News Updates] [RFE/RL]The Azadi Briefing: Taliban Deals Another Blow To Afghan Media[Pakistan News Updates]](https://gdb.rferl.org/0bd80000-0aff-0242-45f1-08d9add418ac_cx0_cy5_cw0_w800_h450.jpg)
